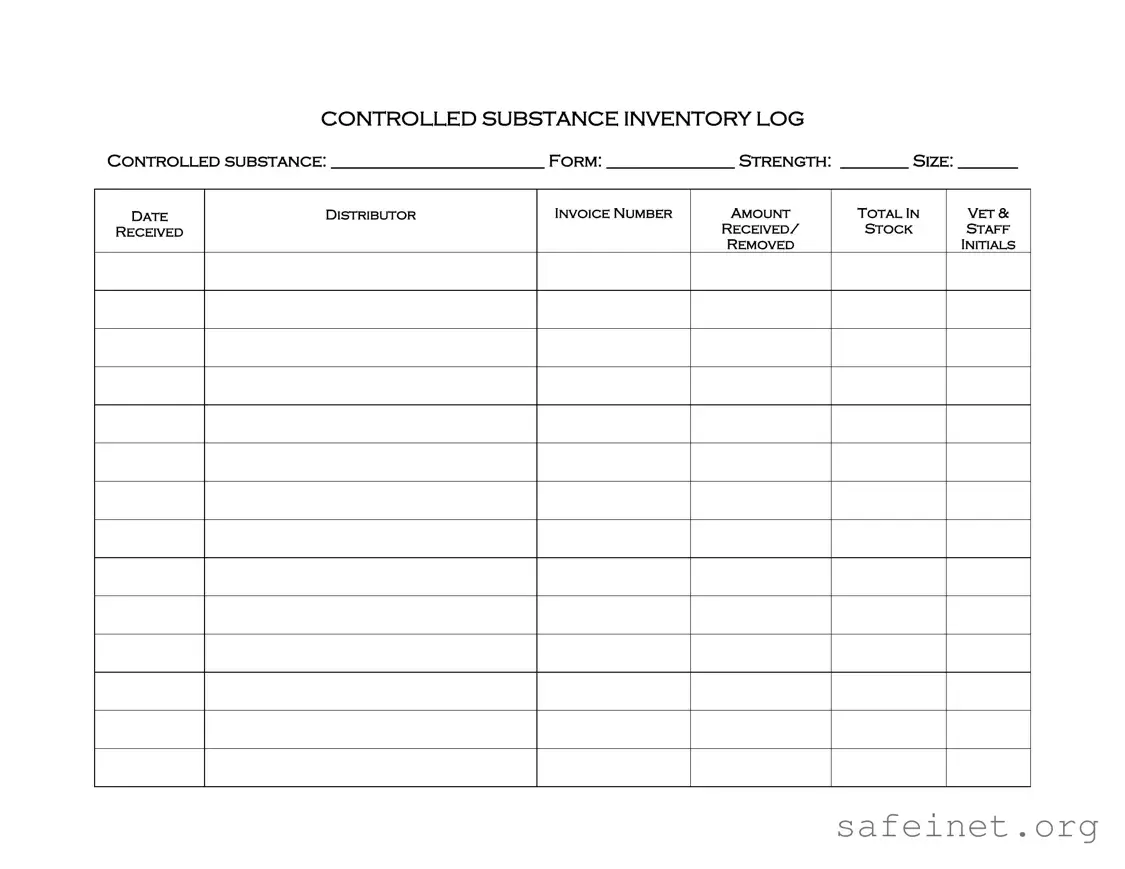
The Controlled Substance Inventory Log serves a similar purpose to the Medication Administration Record (MAR). Both documents track the administration and inventory of medications, ensuring accurate records are kept for patient safety. The MAR focuses on documenting when and how medications are given to patients, while the Controlled Substance Inventory Log emphasizes the management and accountability of controlled substances specifically.
Another document that shares similarities is the Prescription Record. This record details the prescriptions written for patients, including the type of medication, dosage, and quantity. Like the Controlled Drug Log, it requires careful tracking to prevent misuse and to ensure compliance with regulations. Both documents are essential for maintaining the integrity of medication management within healthcare settings.
The Inventory Management Log also aligns closely with the Controlled Drug Log. This log helps organizations keep track of all medications, including controlled substances. It records when stock is received, how much is used, and when it needs to be reordered. Both documents aim to ensure that medication supplies are adequately monitored and maintained.
Similar to the Controlled Substance Inventory Log is the Drug Utilization Review (DUR) report. The DUR evaluates the appropriateness of prescribed medications based on patient history and current prescriptions. While the Controlled Drug Log focuses on inventory management, the DUR assesses the effectiveness and safety of medication use, ensuring that patients receive the best possible care.
The Incident Report can also be compared to the Controlled Substance Inventory Log. This document records any discrepancies or issues related to medication administration or inventory. Both documents are crucial for identifying and addressing potential problems, ensuring that healthcare providers can maintain safe and effective practices.
Another related document is the Medication Reconciliation Form. This form is used during patient transitions, such as hospital admissions or discharges, to ensure that all medications are accounted for. Like the Controlled Drug Log, it helps prevent medication errors and ensures that patients receive the correct medications throughout their care journey.
The Drug Accountability Record is another document that bears resemblance to the Controlled Drug Log. This record tracks the receipt, administration, and disposal of controlled substances within a facility. Both documents serve to enhance accountability and transparency in medication management, reducing the risk of misuse or diversion.
The Supply Chain Management Log also parallels the Controlled Drug Log. This log tracks the flow of medications through a healthcare facility, from ordering to administration. Both documents ensure that proper procedures are followed and that controlled substances are monitored throughout their lifecycle.
The Audit Trail Report is another document that aligns with the Controlled Drug Log. This report provides a detailed history of all transactions involving controlled substances, including who accessed them and when. Both documents are vital for compliance and accountability, helping organizations maintain high standards in medication management.
Lastly, the Compliance Checklist can be compared to the Controlled Drug Log. This checklist is used to ensure that all procedures related to controlled substances are being followed correctly. While the Controlled Drug Log focuses on inventory specifics, the Compliance Checklist provides a broader overview of adherence to regulations and best practices within the organization.